A few things you should know:
What is an ion?
An atom or group of atoms that has a positive or negative electric charge from losing or gaining one or more electrons
An atom or group of atoms that has a positive or negative electric charge from losing or gaining one or more electrons
How an Ion bonds and why?
An Ion bonds to gain a full level of valence electrons or an even number, for stability. An Ion bonds by giving up one or more electrons or by gaining one or more electrons.
An Ion bonds to gain a full level of valence electrons or an even number, for stability. An Ion bonds by giving up one or more electrons or by gaining one or more electrons.
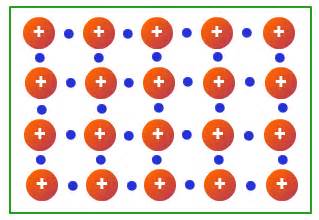
Mitalic bonding - the type of chemical bond between atoms in a metallic element, formed by the valence electrons moving freely through the metal lattice.
Electron Sea Model
To start you will need this:
Naming compounds/ Nomenclature
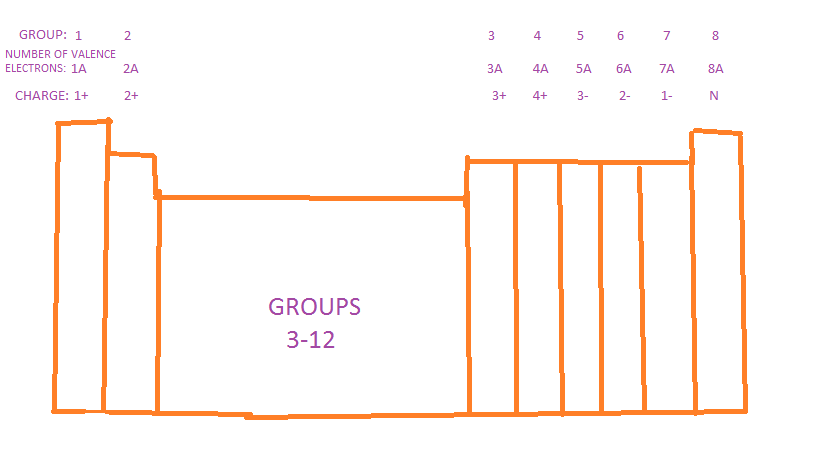
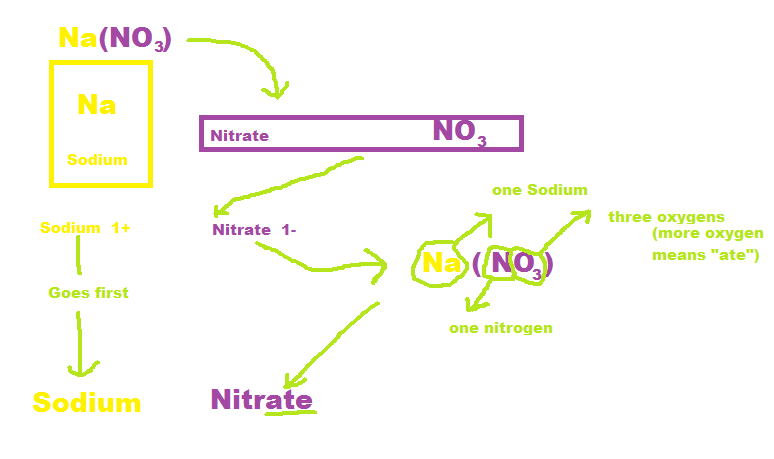
1) First you find the names of the symbols in the formula, using your periodic table. Also find the elements charge.
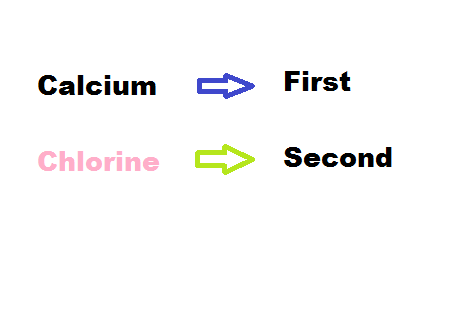
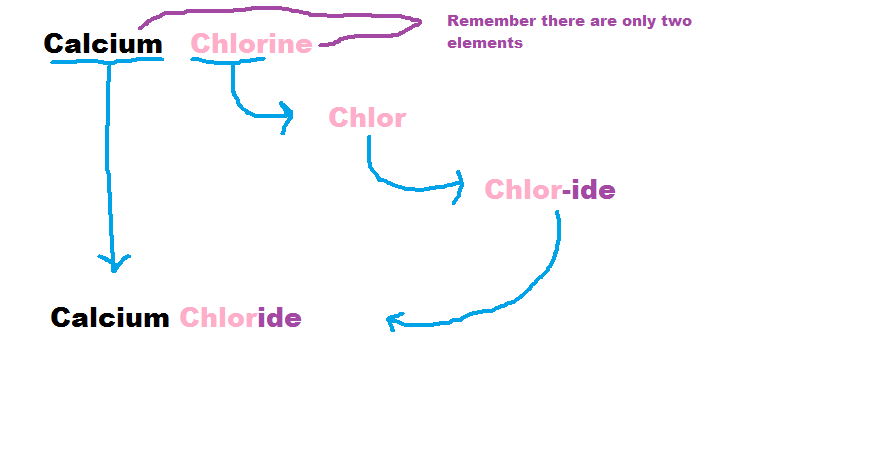
2) Rearrange the names so that the positive element is first.
3) Take the full name of the first element to make the first word.
4) Then using the root of the remaining element you use the ending "ide"
4) Then using the root of the remaining element you use the ending "ide"
5) If you are dealing with three or more elements depending on the amount of oxygen you use the endings "ate" (For when there is more oxygen present than the other elements) and "ite" (for when there is less oxygen present in the formula).
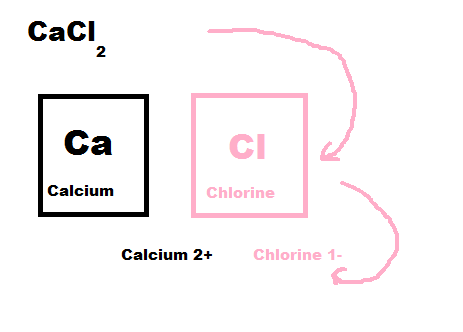
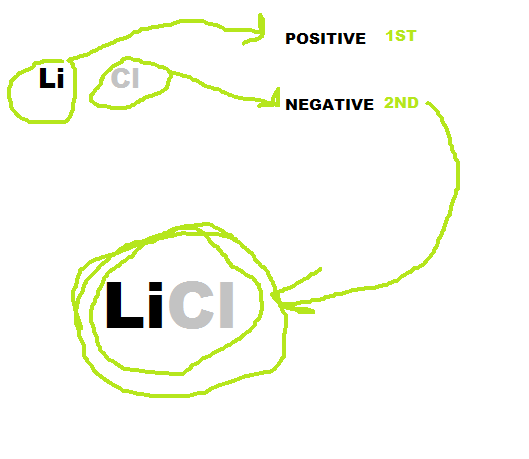
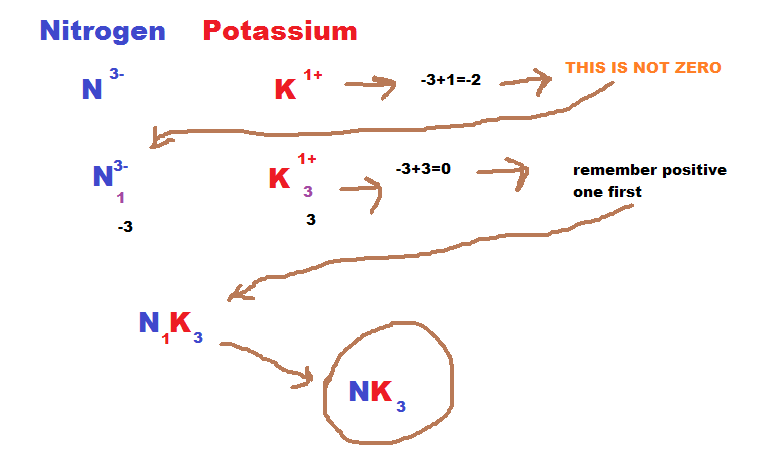
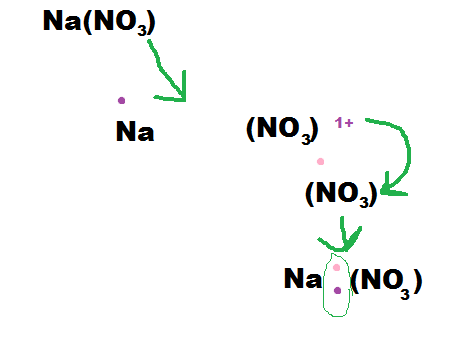
How to write the formula:
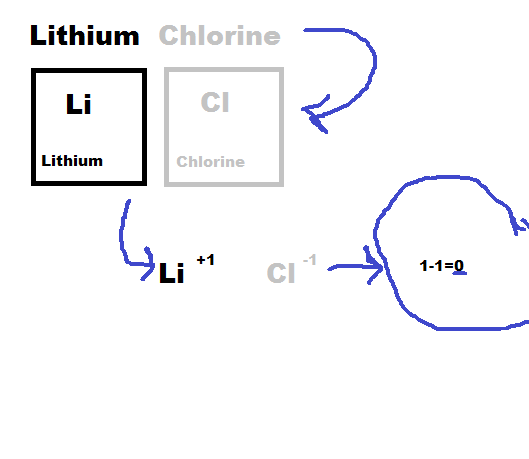
1) First you find the elements symbol and charge, then you add together the two charges. If they equal out then you move to step 2) if not then move to step 3).
2) If the charges equal then put them together with the positive element first.
3) If the charges are not equal then you take the number of the charge and cross it over. Then you multiply them, and if they equal out then you move the number of the first element to the second element and the first elements number to the second element.
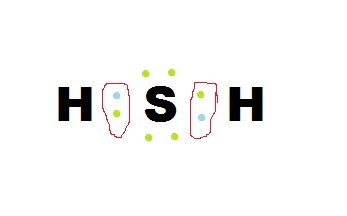
How to form the Lewis Dot Structure:
1) Using the periodic table, determine the number of valence electrons their are for each element involved and write their Lewis Dot Structures individually.
2) Then put them together like a puzzle. (there are many ways to accomplish this so don't freak if you have something different)
3) For special cases that are not on the periodic table, use their charge number as that elements valence electrons.
WORK CITED: www.merriam-webster.com/dictionary/ion http://dictionary.reference.com/browse/metallic-bond