Greek Philosophers:
They believed that all elements were contained in an object, and only went off of what they could observe. For example when they burned wood they believed they were letting the fire out of the wood.
This was the general thought for a long period of time, it was the basis of our knowledge before thoughts of the atom even appeared.
Democritus:
All things are composed of atomos that are mostly comprised of empty space.
Democritus's ideas were not excepted and were completely rejected by the general populace because he could not answer what held them together. Along with that, his ideas were opposed a well known philosopher Aristotle. As such his ideas were lost for around 2000 years.
John Dalton:
The atom was the was an extremely small particle that is spherical, and indivisible. It makes up everything, and can be neither created nor destroyed.
This was a great advance in our knowledge of what makes up stuff. We now knew that objects were composed of small particles and this helped us understand these objects more.
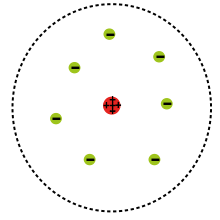
J.J. Thompson:
It was sometimes referred to as the plum pudding model. It was a sphere in shape with negativly charged electrons dispersed evenly throughout it.
Dalton thought that atoms were the smallest particle that made up an object, and Thomson's discovery of the electron proved the existence of subatomic particles.
Ernest Rutherford:
A sphere with a nucleus full of protons. It was surrounded by an electron cloud.
Before Rutherford, the model of the atom had electrons spread out with protons surrounding them. Rutherford was able to show that the atom must not have had protons spread throughout the atom, but instead a small concentrated collection of them: the nucleus. This brought up the idea that the atom was more empty space as opposed to a thick cloud or something as think as plum pudding.
James Chadwick:
A sphere with a nucleus full of protons and neutrons, and then surrounded by an electron cloud.
Scientists soon realized that the newly discovered neutron, as an uncharged but fairly massive particle, could be used to probe other nuclei. It didn’t take long for scientists to find that hitting uranium with neutrons resulted in the fission of the uranium nucleus and the release of incredible amounts of energy, making possible nuclear weapons. Chadwick, whose discovery of the neutron had paved the way for the atomic bomb, worked on the Manhattan Project during WW1.
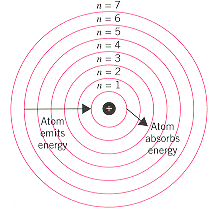
Niels Bohr:
A nucleus filled with neutrons and protons, then surrounded by electron layers.
The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. So opposites don't always attract.
EMC:
The electron cloud is the area around the nucleus of an atom where electrons are most likely to be found.
In later years, the physicists Erwin Schrodinger, Werner Heisenberg and Louis de Broglie pioneered efforts to understand and describe electron behavior. Leading to the the three rules.
Quantum Mechanical Model of the Atom:
3D model of electrons and their relative locations in the atom. There is also a nucleus that has protons and neutrons.
It didn't show the exact location of the electrons but showed there relative locations. Giving us more information about electrons.
Work Cited:
http://abyss.uoregon.edu/~js/glossary/bohr_atom.html science book http://study.com/academy/lesson/electron-cloud-definition-model-theory.html